Wasp forensics

Her home was invaded, larders plundered, and several family members destroyed. All the ingredients for nordic noir fiction.
But this is real life drama and the actors are Australian wasps – far more interesting!
A few months ago we discovered a massive mud nest in the shelter of my parents’ shed in the Clarence Valley, northern NSW. Once carefully prised from their vehicle’s side mirror, the contents caught my attention. A row of cells, each with a cocoon … but not all the same. Something was clearly amiss. Foreign infiltration was my guess.
Underside of massive mud nest after Paul carefully pulled it away from the vehicle’s mirror. Most cells held a shiny, brown cocoon and little else (A, C, D, E, F & G). But in another three (B, H & I), a cellophane-like wrapping separated each golden cocoon from various bits of spider anatomy . And in one cell a grey larva nestled amid a mass of motionless spiders (J). Imaged: 1/1/22
First clue: two types of cocoon
Most of the cells were like Cell A. Simply a shiny, dark brown cocoon suspended by a loose array of white silk threads within in an otherwise empty, ovoid, mud-walled chamber.
But examination of some cells, like B, involved a more archaeological dig. A tangle of spider remains pushed up against one wall. Then a fragile, transparent film lining the entire cell. Beneath that, at one end of the cell, a fine mesh of golden silk supported a tightly woven cocoon of the same material.
Cells A & B, before nest was dismantled.
Imaged: 1/1/22
And there was one more piece to the puzzle.
Immediately alongside the large mud nest was a second blob. Same general appearance, just much smaller. So we popped it off too and included it in the evidence box (labelled Cell K). The spiders packed within were visible through the broken wall of the cell.
Underside of second, smaller mud nest found alongside the main one.
Imaged: 1/1/22
Identifying victim and culprit was easy enough. Piecing together the full detail and sequence of events, however, required more time and detective work. First secure the scene, then isolate the various elements. Examine and record the evidence. And finally compare the findings with what is known from other cases.
Forensic science, indeed!
The case of Sceliphron v Chrysis
So now, four months on and the investigation complete, allow me to present my case (with supporting evidence in italics).
1. The massive mud nest is the work of a lone female Sceliphron
She hatches in the summer, a lone grub within a small dark cell. After feasting on the stunned spiders that her mother left for her, she spins a cocoon … and then waits out the winter months of 2021. In spring, her transformation begins – from a formless yellow sac to an inch-long, slender wasp with large eyes, long legs, wings and strong mandibles. A brief rest to recover from the exertions of her final moult, and then she chews her way free of her thick-walled enclosure.
Out in the late November sun, she feeds at flowers and pairs with a smaller male. Having successfully mated, she flies off alone in search of a suitable site to build a nest. She is seeking a dimly-lit, sheltered, and undisturbed location. A rocky overhang or hollow tree will do, but closer by and easier to find are human constructions – just like the building her own mother chose last summer!
It is now early December 2021 and she has found the perfect place. A smooth, vertical surface inside a dark, uninhabited, weatherproof shed. And with a ready source of water and soil nearby.
Although we never caught sight of her, the mud nest itself provided evidence of the species identity. The location and shape of the nest, with its caches of paralysed orb-weavers spiders, pointed to Sceliphron laetum – a common and widespread species of Australian mud-dauber wasp. The dark brown cocoons also fitted the description for Sceliphron laetum.
Confirmation came when her offspring emerged in February 2022. Sceliphron laetum – proof positive.
Once I knew the species identity, I was able to deduce much about her life. For example, most Sceliphron laetum larvae overwinter in the prepupal stage (Smith, 1979). And nests are more common in and around buildings than they are in natural environments. Therefore it is highly likely that the nest builder spent her own larval life in a nearby building, from an egg laid in early 2021.
The timing of nest establishment I calculated using the emergence dates of the young and the female wasp’s narrow window of opportunity. She could not have started before mid November 2021. Here I call Mum and Dad as witnesses. They last drove their motorhome on 15 November 2021 – and if the nest was there then they would certainly have noticed!
2. She adds a second storey early on
Throughout December, Sceliphron works on her nest. Hauling mud from various sources of wet soil nearby – one jawful at a time – she builds a series of cells. With a high-pitched buzzing, she glues each new blob to the growing structure. It is hard work. Each cell requires a couple of hours of mud-collection, around 30 round trips. And if she finishes a cell before dark, there is still more work to do before taking a break.
When a cell is complete, she goes hunting. Her prey? Orb-weaving spiders. She paralyses each victim before carrying it back to her nest. Once inside the newly built cell, she purposefully positions the motionless spider and attaches a single, large egg to the spider’s abdomen. It takes only minutes* for her to pack the cell with more paralysed spiders, before sealing the opening with a few final loads of mud.
Only then can she rest and seek food for herself. Nectar from flowers provides the energy she needs before tackling construction of the next cell.
After attaching the first few cells to the smooth substrate, she adds the next three on top.
Periodically she covers the entire cluster of cells with extra layers of mud – further protection for her developing brood. And then she continues to add more cells, in two layers.
*The rate of prey collection in the closely related species Sceliphron formosum has been timed at just 2-5 minutes per spider (Callan, 1988).
The changes in mud colour and the sequence of emergence of the young demonstrate the pattern of construction. Why does it matter? Because it tells us much about the behaviour of the invader (details of the crime described below).
3. Very early on, she is robbed by a cuckoo wasp
During the first stage of building, and before the industrious Sceliphron seals the opening of Cell B, an intruder sneaks in. A brilliant blue, female cuckoo wasp – Chrysis lincea.
Unlike Sceliphron, the cuckoo wasp lacks a venomous sting. However, she has little to fear from the much larger Sceliphron. The sculptured cuticle of Chrysis is particularly thick, and she can curl her body into a protective ball should she come under attack.
Inside Cell B, Chrysis lays a single egg and then departs before Sceliphron closes the door. The invader’s egg soon hatches. The little Chrysis grub destroys the unhatched Sceliphron egg – then settles in to feed on the stash of paralysed spiders.
Chrysis lincea is more thief than murderer. The host egg is killed, it is true, but the invader’s prime objective is to steal the living space and the food cache.
Such theft frees the adult Chrysis of both house-building and hunting responsibilities. She has little to do but seek out another suitable cell and lay another egg. In contrast, the nesting Sceliphron female labours on for weeks, presumably unaware of her loss.
In late January, when I dissected the nest to examine the cells and isolate their contents, I found that the cocoon in Cell B contained a well-developed, colourful inhabitant. Through the cocoon wall I could see it kicking and wriggling. So I was not surprised when it broke free the very next day.
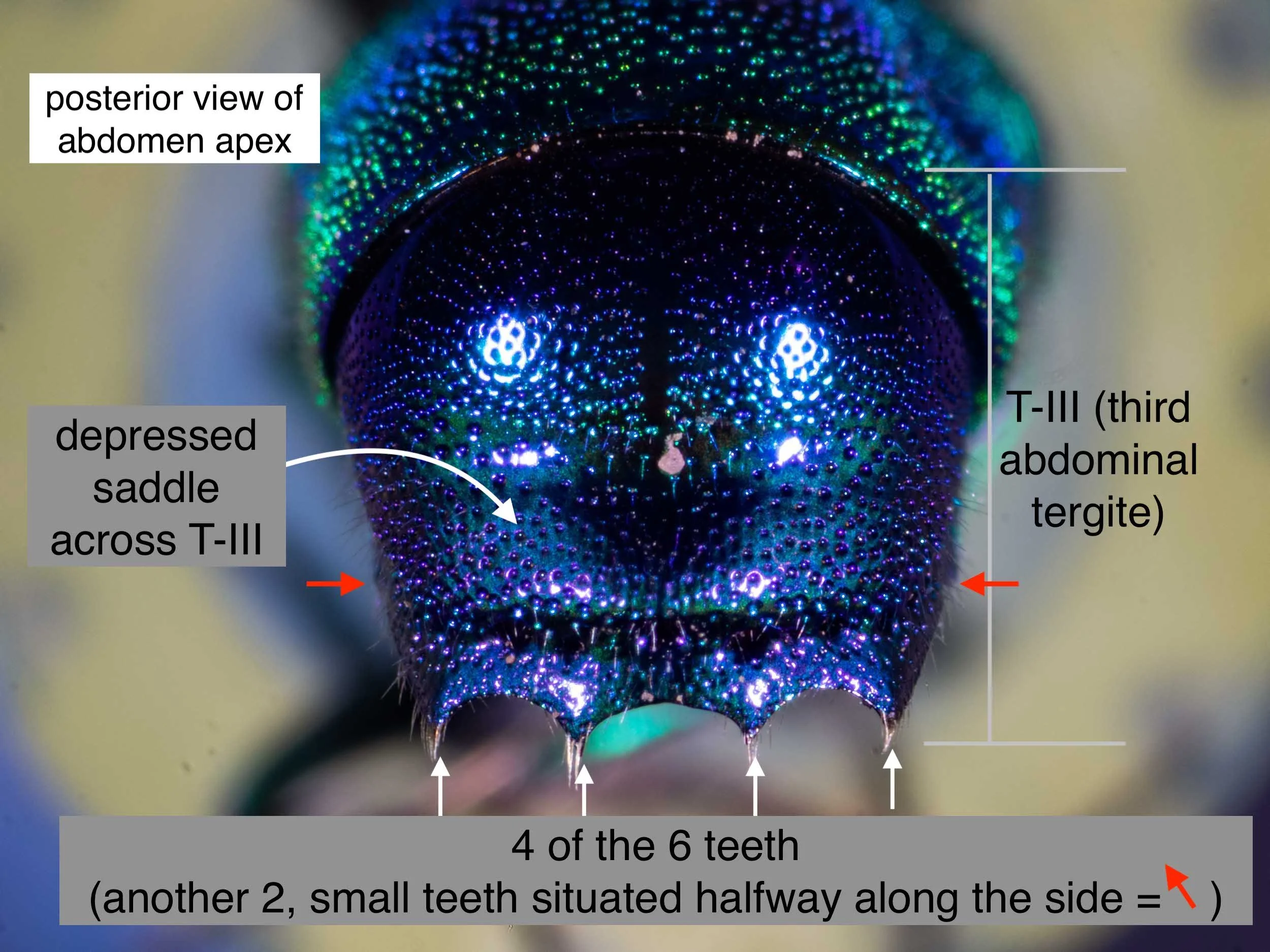
By the wasp’s shape and brilliant, sculptured cuticle I knew it for a cuckoo wasp – family Chrysididae, subfamily Chrysidinae. I ruled out the smaller genera (Stilbum, Primeuchroeus and Praestochrysis) based on the structure of the abdomen apex. This left Chrysis, which is by far the largest genus in Australia (~25 species).
A closer look (plus a bit more reading) and I was sure. Chrysis lincea, one of Australia’s most common cuckoo wasps.
And yes, ‘Chrysis B’ is dead in the panel of three images above. Early on in the investigation, before transporting the nest south with us, we resolved not to release any of the nest inhabitants. We didn’t collect the nest in our home forest and we’ve never seen either species here locally. And we couldn’t realistically return them to the Clarence Valley. It is a 1300km road trip each way! So instead, I preserved the entire collection. Who knows? They may be of value to a wasp researcher someday.
4. Later, a second invasion by Chrysis
It is mid December before Chrysis strikes again. The nest is now much larger. A dozen cells in two tiers, the entire structure well covered by dried mud. Sceliphron is up to the 4th phase of her building work. And nearby, a little blue wasp watches and waits.
Over the following week, as each new cell nears completion and is loaded with spiders, the little thief invades and deposits a single egg. And so Sceliphron loses possession of another four cells. She carries on regardless, sealing the door of each stocked chamber in turn – but too late to keep her own offspring safe.
Are we looking at a serial offender, or are multiple Chrysis involved? It is not possible to know for sure.
The last 4 cells added to the main nest contained cuckoo wasps, and all were identical to Cell B. So they are all the same species – Chrysis lincea.
The original female Chrysis may have returned, or perhaps a different individual is responsible. It would not be difficult for wandering Chrysis females to spot Sceliphron gathering mud and to follow her back to the nest.
5. She builds one final cell – an insurance policy
Sceliphron has noticed Chrysis hanging about, and it troubles her. She adds layer after layer to the main nest, seeking to protect her brood. As a final strategy, she starts a new nest … and late in the day, when Chrysis is less active. She chooses only large spiders this time, and so needs make only 3 trips before sealing the chamber. The new nest, with its single cell, is quickly encased in a thick mud wall of its own.
And that’s it. She’s done. With 18 cells (plus 1), her nest is larger than average for her species*.
It’s now 5 weeks since she emerged as an adult, and she doesn’t live to see in the new year**.
OK, I am now getting more speculative – at least in regard to any intent on the part of Sceliphron. But there is a basis for my hypothetical.
One species of European sphecid wasp has been shown to respond to the presence of cuckoo wasps by shifting its nesting activity to later in the day (Strohm et al., 2001).
And there is definitely something unusual about that final cell, the one I labelled Cell K.
I’m confident it is a Sceliphron – both the larva and its cocoon are identical to the other Sceliphron. But the progress of development has been strikingly different. It reached the prepupal stage … but then nothing. No transformation into a pupa, no decomposition in death. No change. For 3 months now!
I am quite confident that the larva from Cell K is now in diapause. Indeed, most Sceliphron laetum larvae spend around 9 months as prepupae, emerging the following summer (Smith, 1979).
If I’m correct, and if nothing goes awry with the naked prepupa, development of ‘Sceliphron K’ will start up again in Spring and the adult will emerge in early summer. I will be watching, and I am patient.
Construction of this nest was a lot of work for one little wasp. Completed, the main nest was 14cm long with a dry weight of 130g. Such an investment apparently pays off in the high survival rate of her offspring. Smith (1979) reported an “extremely low larval and pupal mortality” for Sceliphron laetum (p. 181). Interestingly, the same author demonstrated that attack by Chrysis was one of the main causes of loss.
*Sceliphron laetum are solitary insects and, on average, build nests with 8 cells (Smith, 1979).
**An adult female Sceliphron laetum may live for up to 6 weeks and build more than one nest (Smith, 1979).
And so I rest my case.
Well, almost.
In gathering the evidence cited above, I amassed a wealth of images and learned much about the development of Chrysis lincea.
Below are four findings, just for the record.
Chrysis lincea builds a compound, covered cocoon
The cocoon of Chrysis lincea is unusual, and no simple structure.
It comprises three distinct layers.
The best way to explain is to take a look at the real thing.
This style of cocoon is unusual … but not unheard of. Most notably, covered cocoons were described for four Chrysis species in a review of cocoons from chrysidid wasps of Eastern Europe (Martynova, 2020).
It is the outer ‘covering’ that makes this a special kind of cocoon. That brittle, thin layer of fibrous silk sits close to the host cell wall, and might be mistaken for a lining of the host cell. But it is produced by the cuckoo wasp larva, not the nest builder or her offspring. Martynova (2020) assigns the outer layer two functions – to “separate the remains of provisions and fixate the cocoon body inside the host cell” (p. 131). That certainly fits with what I see in Chrysis lincea.
Empty cocoon, shortly after the emergence of Chrysis adult (H) on 9 Feb, 2022
Once fully developed, the wasp cuts or tears away a circular piece from the end of the golden cocoon, pushing aside both the inner and middle layers. It then has space to move, and perhaps rest, before tackling the dried mud of the host cell wall at the ‘head’ end of the cell. Of course, the thin outer covering of its own cocoon presents no great challenge.
2. Chrysis lincea is a kleptoparasite
There are two main strategies adopted by the nest parasites of wasps.
A - hatch early, destroy the host egg, and consume the host’s food.
B - hatch or invade late, and consume the well-fed host at its prepupal or pupal stage (Martynova & Fateryga, 2015).
Like most Chrysis species, C. lincea is a Type A parasite.
This became apparent early on in the investigation of the nest. For the record, I present three independent lines of supporting evidence.
First, there are the spider remains.
Those in the Chrysis-occupied cells are recognisable as orb-weavers. Most have intact head-thorax and legs. In many, the abdomen is also still attached – albeit collapsed like a burst balloon.
In contrast, I rarely found more than a fang or leg fragment in the Sceliphron cells.
There is a difference in the spider remnants, depending upon the wasp species occupying the cell.
Second, the Chrysis cocoon is not surrounded by the host cocoon. A Type B parasite feeds inside the host cocoon, and therefore spins its own cocoon inside that of the host (Martynova & Fateryga, 2015). I found no sign of a host cocoon in any of the cells taken over by Chrysis lincea.
Third, I have direct evidence! The wasp grub in Cell J was clearly feeding on spiders. After transferring the entire contents of Cell J to a dish, I watched on as the larva fed voraciously and grew rapidly.
A few days later, the Chrysis larva began to wander the dish. It pushed the spider remains about as it lay down fine, white silk. On 8th January it settled and started to produce the golden silk of the cocoon.
The Chrysis larva selectively devoured the soft abdomen of each spider. This resulted in rather a lot of leftovers! Normally, within the cell, the spider remains would have been pushed up against the host wall as the larva lay down the outer covering of its cocoon.
3. Egg to adult in eight weeks: the developmental stages of Chrysis lincea
The wasp in Cell J was only a few days old when we discovered the nest. I decided to monitor and document its development in detail, first by removing the larva from the cell, and later removing the pupa from the cocoon.
It was while studying this individual – ‘Chrysis J’ – that I became fascinated by the details of cuticle and colour formation. That digression ultimately led me to write a completely separate blog post (A bright, new skin), in which ‘Chrysis J’ is again a lead actor.
Here is a summary of the morphological transformation of ‘Chrysis J’, week by week.
Week 1: hatching; feeding; growing and moulting
Week 2: final feeding and growth; moults to (non-feeding) prepupa; spins cocoon
Weeks 3-5: prepupa moults to pupa; final moult to adult begins (but all largely out of sight)
Week 6: sclerotization of the adult cuticle beneath the pupal cuticle; refinement of appendages
There are so many, rapid changes to the cuticle during Week 6 that it warrants a closer look.
The wasp’s underbelly is the last change. The ventral plates of the abdomen remain soft and pale long after the dorsal plates, thorax and head have become fully coloured and hardened.
Week 7: pupal cuticle shed; wings expand; wriggles and kicks
Week 8: increasingly active and responsive; freely moving and preening within a few days
By 14 Feb ‘Chrysis J’ was twitching near-continuously, standing and almost walking, but with wings still askew. When I checked early the next morning it was walking, albeit unsteadily, and the wings were aligned. It was not until 16 Feb that it was freely running around the dish.
Note: The apparent change in colour from the blue of Week 7 to the green of Week 8 is not real. It’s simply because I used a different light source for the photos.
4. The final moult in Chrysis lincea is a protracted affair
Moulting involves much more than shedding of the old cuticle. Formation of the new cuticle starts long before and continues for some time after the insect frees itself from the skin of its earlier stage. I go into details – with Chrysis as an example – in A bright, new skin.
There is one aspect that particularly surprised me. After Chrysis sheds its pupal cuticle and the wings expand, there is still a week before the abdomen assumes its final shape and the insect is able to move freely.
When I first saw this happen with ‘Chrysis I’, I was alarmed. Had something gone wrong? Did my removal of the cocoon lead to this deformity? But eventually the wasp became strong and active.
Later, I watched on as the development of ‘Chrysis J’ followed the exact same pattern. This protracted step in the final moult corresponds to Step 5 in the moulting schema described in A bright, new skin.
Lying defenceless, wings spread and abdomen soft, these wasps seem terribly vulnerable. But normally this entire process takes place within the cocoon. Even when the wasp breaks free of the golden silk capsule, it remains safe inside the outer envelope and the host mud cell. Eventually, sometime in Week 8, it is ready to face the outside world.
And finally, a few additional image galleries.
Just for those of us who are more than a little bit obsessed by wasps :)
Rogues’ gallery
Host’s family album
The L files
Discovering the published work of Dr Kate Martynova was a real boon for me. Her research includes the life history, cocoon development, ecology and taxonomy of cuckoo wasps.
Completing her university studies in Donetsk, Kate is now based at the National Academy of Sciences, Kyiv.
What must her life be like right now?
We can only hope that she and her colleagues are safe, and soon free to continue their lives and wasp studies in Ukraine – in peace.
References
Bohart, R. M. 1985. A key to the Australian Chrysis with description of new species (Hymenoptera: Chrysididae). Insecta Mundi, 1(1), 47-52
Callan, E. McC. 1988. Biological observations on the mud-dauber wasp Sceliphron formosum (F. Smith) (Hymenoptera: Sphecidae). The Australian Entomologist, 14(6), 78-82.
Elgar, M.A. & Jebb, M. 1999. Nest provisioning in the mud-dauber wasp Sceliphron laetum (F. Smith): Body mass and taxa specific prey selection. Behaviour, 136: 147-159
Martynova, K.V. & Fateryga, A.V. 2015. Chrysidid wasps (Hymenoptera, Chrysididae) – parasites of Eumenine wasps (Hymenoptera, Vespidae: Eumeninae) in Crimea. Entomological Review, 95, 472-485
Martynova, K.V. 2020. Cocoons of cuckoo wasps (Hymenoptera: Chrysididae): First overview of morphology reveals unexpected traces of taxonomy. Zoologischer Anzeiger, 285, 122-138.
Smith, A. 1979. Life strategy and mortality factors of Sceliphron laetum (Smith) (Hymenoptera: Sphecidae) in Australia. Australian Journal of Ecology, 4: 181-186
Strohm, E., Laurien-Kehnen, C. & Bordon, S. 2001. Escape from parasitism: spatial and temporal strategies of a sphecid wasp against a specialised cuckoo wasp. Oecologia, 129, 50=57
unknown auther (perhaps Houston, T). Cuckoo Wasps, Western Australian Museum Factsheet, last updated 2013. Accessed 10/5/22 - link